T39.1
DESCRIPTION
Poisoning due to paracetamol by adolescents is generally due to intentional ingestion. The accidental ingestion of paracetamol elixir preparations by toddlers very rarely causes toxicity. Toxicity can be due to acute ingestions or repeated supratherapeutic ingestion (RSTI). Toxicity due to IV paracetamol may also occur.
High-risk patients (glutathione deficiency, liver disease, use of enzyme-inducing drugs, patients with recent illness or dehydration) may experience toxicity at lower doses.
DIAGNOSTIC CRITERIA
- An acute ingestion in excess of 150 mg/kg per 24-hour period in healthy children is potentially toxic.
- Serum paracetamol concentration must be measured at least four hours following ingestion.
- Use nomogram to assess risk of toxicity.
Paracetamol treatment nomogram
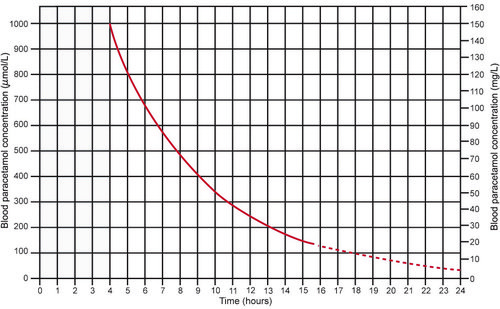
Source: Daly FF, Fountain JS, Murray L, Graudins A, Buckley NA; Panel of Australian and New Zealand clinical toxicologists. Guidelines for the management of paracetamol poisoning in Australia and New Zealand -explanation and elaboration. A consensus statement from clinical toxicologists consulting to the Australasian poisons information centres. Med J Aust. 2008 Mar 3;188(5):296-301.
- Cautions for use of this chart:
- The time co-ordinates refer to time since ingestion.
- Serum levels drawn before 4 hours may not represent peak levels.
- Use the graph only in relation to a single acute ingestion.
- Do not use when there is a history of RSTI, a history of ingestion of extended release formulation or delayed presentation (> 24 hours post-ingestion).
Repeated supratherapeutic ingestions
RSTI defined as:
- > 200 mg/kg or 10 g (whichever is less) over a single 24-hour period.
- > 150 mg/kg or 6 g (whichever is less) per 24-hour period for the preceding 48 hours.
- > 100 mg/kg or 4 g/day (whichever is less) in patients with predisposing risk factors.
This nomogram is not designed for use in RSTI.
Investigations
If toxic dose ingested or patient symptomatic, do:
- Serum paracetamol level
- Baseline urine and electrolytes
- ALT
- INR
GENERAL AND SUPPORTIVE MEASURES
- Gastric lavage and activated charcoal, see Poisoning, Treatment .
MEDICINE TREATMENT
Acute ingestion
- For acute ingestion, initiate treatment with N-acetyl cysteine (NAC) if the blood paracetamol concentration for the time since ingestion falls to the right of the curved line on the nomogram.
- Administer without waiting for plasma paracetamol levels in substantial overdose, defined as ≥ 10 g (20 tablets) or ≥ 150 mg/kg, whichever is smaller. Discontinue if plasma levels are in the non-toxic range.
- If patients present > 8 hours post-ingestion AND has taken a potentially toxic dose, start on NAC without waiting for the paracetamol levels.
- If the time of ingestion is unknown, start treatment for any detectable level of paracetamol or any elevation of AST or ALT.
Repeated supratherapeutic ingestions (RSTI)
Obtain a second paracetamol level and ALT 4–6 hours after the original 4 hour concentration. If either second paracetamol (above the treatment line) or ALT are abnormal, proceed with NAC infusion and repeat serum paracetamol level and ALT after 8 hours.
N-Acetylcysteine, IV.
First 24 hours
- Loading dose: 150 mg/kg in dextrose 5%, 5 mL/kg given over 1 hour.
- 50 mg/kg in dextrose 5%, 5 mL/kg over the next 4 hours; then 100 mg/kg in dextrose 5%, 10 mL/kg over 16 hours.
Second 24 hours
- 100 mg/kg in dextrose 5%, 10 mL/kg over 24 hours.
REFERRAL
Patients with severe hepatotoxicity as indicated by any of the following:
- INR > 2 at 24 hours or > 3 at any time after overdose
- pH < 7.3 or bicarbonate < 18 mmol/L
- hypovolaemia
- encephalopathy
- creatinine > 200 µmol/L